Articles
- Page Path
- HOME > Res Community Public Health Nurs > Volume 34(3); 2023 > Article
-
Original Article
- Gender Differences in Older Adults’ Muscle Strength and Depressive Symptoms: A Relationship Mediated Through Perceived Stress
-
Gyeong A Kang1
 , Jihye Shin2
, Jihye Shin2 , Ju Young Yoon3
, Ju Young Yoon3
-
Research in Community and Public Health Nursing 2023;34(3):196-204.
DOI: https://doi.org/10.12799/rcphn.2023.00164
Published online: September 27, 2023
1Ph.D. student, College of Nursing, Seoul National University∙Center for Human-Caring Nurse Leaders for the Future by Brain Korea 21 (BK 21) four project, College of Nursing, Seoul National University, Seoul, Korea
2Ph.D. student, College of Nursing, Seoul National University, Seoul, Korea
3Professor, College of Nursing, Seoul National University∙Center for Human-Caring Nurse Leaders for the Future by Brain Korea 21 (BK 21) four project∙Research Institute of Nursing Science, Seoul National University, Seoul, Korea
- Corresponding author: Ju Young Yoon PhD, RN, Professor, College of Nursing and Research Institute of Nursing Science, Seoul National University, 103 Daehak-ro, Jongno-gu, Seoul 03080, South Korea Phone: +82-2-740-8817, Fax: +82-2-741-5244, Email: yoon26@snu.ac.kr
Copyright © 2023 Korean Academy of Community Health Nursing
This is an Open Access article distributed under the terms of the Creative Commons Attribution NoDerivs License. (http://creativecommons.org/licenses/by-nd/4.0) which allows readers to disseminate and reuse the article, as well as share and reuse the scientific material. It does not permit the creation of derivative works without specific permission.
- 1,123 Views
- 48 Download
Abstract
-
Purpose
- Preventing the effects of physical vulnerability is a practical approach to improving older adults’ depressive symptoms. This study aims to examine the relationship between gender differences related to muscle strength and depressive symptoms mediated by perceived stress.
-
Methods
- In this cross-sectional study, data from 2,705 older adults (65 years old or older) from the Korea National Health and Nutrition Examination Survey VII 2016 and 2018 were analyzed. The moderated mediation model was developed; the outcome, independent, mediation, and moderated mediation variables in the literature-based research model were depressive symptoms, muscle strength, perceived stress, and gender, respectively.
-
Results
- Perceived stress had a mediating effect on the relationship between muscle strength and depressive symptoms. The indirect effect of muscle strength on depressive symptoms mediated by perceived stress was β = -.02 (95% CI:-0.03~-0.01). The moderated mediation model demonstrated that the interaction term of handgrip strength and gender negatively affected perceived stress, which indicated that gender moderated the mediating model of perceived stress on the association of muscle strength and depressive symptoms (β = -.01, p <.05). The conditional indirect effect model was insignificant in the male group (β = -.00, 95% CI:-0.01~0.01) but significant in the female group (β = -.01, 95% CI: -0.02~0.00).
-
Conclusions
- Perceived stress mediated the relationship between muscle strength and depressive symptoms. However, the effect differed by gender. A stress-mediated depressive symptoms intervention program for older adults should be developed to consider women’s needs.
- Depressive symptoms are prevalent in older adults and a severe public health concern among older adults [1]. A recent meta-analysis has shown that depressive symptoms have increased fourfold among frail people compared to non-frail people [2]. Using muscle strength as one of the important indicators, frailty constitutes a physically vulnerable status and is closely related with depressive symptoms [3-5]. Likewise, older adults are prone to physical limitations, which induce feelings of helplessness [6,7]. The factors mediating the influence of older adults’ low muscle strength on depressive symptoms must be elucidated for efficient intervention. Therefore, in this care area, ameliorating the effect of low muscle strength is a practical approach to improving depressive symptoms among older adults.
- Owing to their physical limitations and lower capacity to manage external stimuli, physically vulnerable older adults may experience higher stress levels and depressive symptoms compared to their robust counterparts [2,4]. Lazarus and Folkman [8] defined “stress” as a relationship between an individual and their environment that is appraised as personally significant and taxing or exceeding one’s resources to cope; they also highlighted individual differences in stress response according to appraisals of stressful situations and the resources available for managing stress. When frail people are exposed to stressful situations, they experience greater stress. This is because their vulnerable physical status restricts their daily life and causes more discomfort [4], manifesting as decreased motor function and quality of life [9]. Physical activity also affects the release of neurotransmitters and myokines, which are significantly associated with stress response [10].
- Stress is significantly associated with depressive symptoms [7,11]. Older adults experience various stressful situations owing to decreased muscle strength [6,7]. Moreover, the degree of stress is associated with the intensity and duration of stressful life events and the ability to manage them. Smith et al. [12] examined the relationship between muscle strength, depressive symptoms, and stress; however, in their study, gender was simply considered a confounder. Additionally, many studies have examined the relationships between muscle strength, depressive symptoms, and stress [3,4,7,11]. However, research on the relationship between the three variables, namely, muscle strength, depressive symptoms, and stress is scant. Therefore, there is a need for further research to understand the relationship more accurately.
- Depressive symptoms show gender differences; females show more severe depressive symptoms than males [13]. The factors causing gender differences can be considered in endogenous and exogenous aspects. As endogenous aspects, females’ peak muscle mass is lower than males’ [14,15]. Thus, female older adults reach frailty status and are exposed to situations that may limit their daily lives earlier than males [5]. One study showed greater activation in the Hypothalamic-Pituitary-Adrenal (HPA) axis and autonomic nervous system during stress response in the male group compared to the female group [16]. As for exogenous aspects, scholars point to socio-cultural factors. Females experience chronic stress by taking caregiver roles in most families [17]. In addition, the unfairness of education and social participation opportunities between the genders in the past have contributed to today’s economic inequality [18]. Sufficient socioeconomic factors act as a buffer for stress, but females’ capacity for coping with stress tends to be weak, caused by accumulated external factors.
- Considering previous studies, gender differences must be understood in terms of the relationship between physical and psychological vulnerability. Therefore, this study aimed to examine the gender differences concerning muscle strength and depressive symptoms mediated by perceived stress by using data from the Korea National Health and Nutrition Examination Survey (KNHANES) VII.
Introduction
- 1. Study Design and Participants
- This study is a cross-sectional study. The data for this study were drawn from the 2016 and 2018 waves of KNHANES VII, which were obtained from the Korea Disease Control and Prevention Agency (https://knhanes.kdca.go.kr/). The KNHANES is a national surveillance system that represents the Korean people's health status. More details on the data collection process are available [19]. From the available data, participants aged 65 years or older were selected. Among them, those with physical disabilities owing to dementia or with missing values for grip strength, perceived stress, and depressive symptoms were excluded. Ultimately, the data of 2,705 participants were used in the analysis.
- 2. Measures
- Handgrip strength is an independently valuable indicator reflecting physical status and has been included as an indicator in the Cardiovascular Health Study frailty scale [5]. Handgrip strength has been reported to have excellent reliability and validity; it can be measured in healthy participants and those with clinical diseases [20]. When diagnosing sarcopenia, the Asian Working Group for Sarcopenia and the European Working Group on Sarcopenia in Older People used muscle strength via handgrip strength as one of the indicators for screening physical status [21]. We used the handgrip strength variable provided in KNHANES, which was measured with a digital grip dynamometer. The maximum value between the left and right handgrip strength was used for the muscle strength variable.
- Depressive symptom was a dependent variable in our study, assessed with the nine-item Patient Health Questionnaire based on self-reported measures [22]. The total score ranged from 0-27 in a continuous variable, and higher scores indicated higher degrees of depression.
- Perceived stress was a mediating variable of this study. The KNHANES asked participants one question on the degree of perceived stress, to which the responses could be extremely stressed (four points), quite stressed (three points), a little bit stressed (two points), or hardly stressed (one point). A higher score indicated a higher stress level.
- Demographic variables included age (years), residential area (urban vs. rural), education attainment (years), household income (low, middle-low, middle, middle-high, or high), marital status (living with spouse vs. not living with a spouse, widowed, or divorced or never married). Education attainment was categorized as 6 years or less (elementary), 7–9 years (middle school), 10–12 years (high school), and 13 years or more (college). Health-related variables included current smoking (yes vs. no), high-risk drinking (yes vs. no), history of depression treatment (yes vs. no), and the number of chronic diseases [12,23]. Specifically, high-risk drinking refers to having more than seven glasses of alcohol twice a week for males and drinking more than five glasses twice a week for females. Given that treatment for depression could affect the degree of depression, it was included as a covariate. Regarding the number of chronic diseases, participants were asked whether they have been diagnosed with the following 10 kinds of chronic diseases by a physician: hypertension, osteoarthritis, diabetes, cerebrovascular disease, cardiomyopathy, angina, asthma, kidney disease, chronic obstructive lung disease, and cancer (colon, stomach, liver, breast, cervical, lung, thyroid, and other cancers). Each item was allocated 1 for “yes” and 0 for “no,” and a higher total score indicated having more chronic diseases (Table 1).
- 3. Statistical Analysis
- We conducted a complex sample analysis by applying colonies, stratification variables, and the weights of the 2016 and 2018 data. First, we derived descriptive statistics to show the general characteristics. Second, we performed a correlation analysis between variables, including the main variables and covariates. Third, we confirmed a simple mediation model of stress among all participants. Fourth, we confirmed the moderated mediation model adjusted by gender as a moderator variable; we used SPSS PROCESS macro, version 3.5.3, developed by Preacher and Hayes, to test the mediation hypothesis. Complex sampling design was not applied in mediation model and moderated mediation model analysis using PROCESS macro.
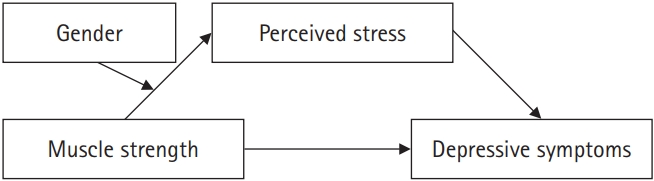
- The moderated mediation analysis was performed based on 5,000 bootstrapped samples using bias-corrected and accelerated 95% confidence intervals (model 7 in PROCESS macro). Mean-centering was used for continuous variables. We then examined the relations among the independent variable X (handgrip strength), dependent variable Y (depressive symptom), mediator M (perceived stress), and moderator W (gender; Figure 1). The conditional process model could be specified using regression equations for M and Y, with the two equations allowing the effect of X to be dependent on W; however, the effect of M on Y to be fixed. Hence, this equation could be modified by including W and XW as predictors to allow the direct effect of X to be moderated by W [24]. Therefore, moderated mediation existed when the following requirements were met: (i) In the regression equation of the mediator, the moderated effect of the moderator between the independent variable and mediator should be significant; (ii) In the regression equation of the independent variable to the dependent variable, the independent variable, and moderator interaction terms should not be significant; (iii) In the regression equation of the independent variable and mediator to the dependent variable, the mediator should be significant [25].
- 4. Ethical considerations
- As secondary data were used in this study, it was exempt from requiring approval by the appropriate institutional review board (IRB No. SNU 21-08-069).
Methods
1) Muscle strength
2) Depressive symptoms
3) Perceived stress
4) Covariates
- 1. Descriptive Analysis
- Table 1 gives the general characteristics of the study population by gender. It includes age, residential area, education attainment, household income, marital status, current smoking, high-risk drinking, history of depression treatment, number of chronic diseases, muscle strength, perceived stress, and depressive symptoms. The correlation between variables showed significant relations between handgrip strength and perceived stress (β = -.07, p <.01), between handgrip strength and depressive symptoms (β = -.19, p <.01), and between perceived stress and depressive symptom (β = .34, p <.01; Table 2).
- 2. Mediating Effect of Perceived Stress on Relations Between Muscle Strength and Depressive Symptoms
- Muscle strength was negatively associated with perceived stress (β = -.01, p <.05) and depressive symptoms (β = -.06, p <.05). The negative effect of muscle strength on depressive symptoms was significant (β = -.04, p <.05) when both muscle strength and perceived stress were concurrently included. The indirect effect of muscle strength on depressive symptoms mediated by perceived stress was β = -.02 (95% CI: -0.03~0.01) (Table 3), indicating the mediating effect of perceived stress on the relationship between muscle strength and depressive symptoms.
- 3. Gender Differences in the Mediation Model of Perceived Stress on Relations Between Muscle Strength and Depressive Symptoms
- We analyzed the relations between muscle strength, perceived stress, and depressive symptoms using gender as a moderator, controlling for demographic and health-related variables among all participants. Table 4 shows that the interaction term of handgrip strength and gender had a negative effect on perceived stress (β = -.01, p <.05). Specifically, the conditional indirect effect model according to gender was not significant in the male group (β = -.00, 95% CI: -0.01~0.01), but was significant in the female group (β = -.01, 95% CI: -0.02~-0.00).
Results
- Effective interventions for alleviating depressive symptoms should be provided to guarantee a good life in old age. Hence, the relationship between physical and psychological vulnerability must be thoroughly understood. Our study examined the relationship between muscle strength and depressive symptoms mediated by perceived stress and identified gender differences in the mediating effect of perceived stress. In particular, the results revealed that the conditional indirect effect model according to gender was not significant in the male group but was significant in the female group. This model explained the results from biological and social perspectives.
- In this study, the mediating effect of perceived stress in the relationship between muscle strength and depressive symptoms was significant. Muscle strength/mass can reflect the quality of muscle [12], and myokines are associated with older adults’ mood [26]. Although the exact mechanism has not been confirmed, it may be assumed that the quality of muscles affects stress response. In the human body, muscles work as endocrine organs. Interleukin-6, a type of myokine released from muscle fibers, is secreted in response to stress and is involved in cortisol secretion in the adrenal gland. Therefore, the circulating level of interleukin-6 is raised by stress [10,26]. In other words, a physically vulnerable state such as muscle weakness leads to greater susceptibility to stress owing to the decrease in endocrine coping function against stress [10]. Long-term exposure to stress causes a chronic stress condition, which increases cortisol resistance and results in depressive symptoms [7,10,12].
- Additionally, physical vulnerability such as low muscle strength status can lead to functional limitations and physical inactivity. Older adults’ vulnerable physical condition can be a contributing factor that induces social isolation, increases perceived stress, and worsens the stress-coping base [27,28]. Increased perceived stress could lead to the development of depressive symptoms [2,3,4]. Physically vulnerable older adults are less likely to engage in social activities. Therefore, it is important to reduce the social isolation attributable to their physical vulnerability [28]. Prevention of muscle weakness should be fundamental. Moreover, additional institutional support to prevent physical vulnerability from acting as a stressor in older adults’ daily lives is crucial.
- The mediating effect of perceived stress differs by gender. The mediating effect of perceived stress was only significant among females in the moderated mediation model of our study. This result indicates that the perceived stress response to decreased muscle strength differs according to gender. This difference can be explained by hormonal responses and sensitivity to stress sources [4,16,29]. First, the physiological system response to stress is mainly explained by responses of the autonomic nervous system and HPA axis [30]. Female gender hormones weaken the sympathoadrenal response and delay cortisol feedback of the HPA axis [16]. Although hormone medication and blood hormone levels were not investigated in the KNHANES, judging from the demographic and health-related variables in this study, the difference by gender can be presumed to be a result of delayed response of myokines or neurotransmitters to stress by gender hormone effects.
- Second, sensitivity to stressors differs by gender. In a previous study, stressors caused sadness or anxiety, which manifested more in physical responses and behaviors in females because females more cognitively focus on sadness or anxiety than males [31]. The environment to which males and females have been exposed also contributes to the differences in sensitivity. Consequently, females tend to consider stressors more threatening [6]. According to a study conducted on twin pairs (male-male, female-female, and male-female) that considered social aspects, females are more sensitive to the depressogenic effects of proximal and distal networking with individuals [32]. The buffer function of interactions with close networks has also been reported in another study [7]. Sensitivity to stress sources can be complemented by the socioeconomic basis of stress management. Gender roles have weakened the stress-coping function of the current female older adult generation [29]. For these study participants, the household income and education attainment gaps between men and women were significant. However, younger male and female generations have experienced similar educational and work environments. Moreover, the socioeconomic gap is less prominent [16,18]. Until the gap is completely bridged, the influence of gender roles accumulated in the past cannot be ignored. In terms of appraising the situation positively and developing coping strategies for stressors [30,33], encouraging social interaction to alleviate stress is expected to be an effective intervention for female older adults’ depressive symptoms.
- With aging, adults experience fluctuating cortisol cycles owing to dysfunction of the HPA axis, inhibiting the proper stress response [16]. Physical activity or financial compensation interventions improve older adults’ psychological health [34]. However, most of them have been implemented without consideration for participants’ gender [12]. Our results highlighted the effect of gender on the pathway of psychological vulnerability among older adults. Even if the biological mechanism for stress response is different according to gender [16,35], gender roles and other external factors play more critical roles in coping with stress [36]. Meanwhile, non-pharmacological interventions (e.g., exercise, psychotherapy, and cognitive behavior therapy) have also been applied to older adults. However, there is a need for further research to confirm the applicability of these interventions in this care area [37]. This study’s results thus suggest practical interventions that may benefit older adults.
- Our study has several limitations. First, the perceived stress variable was measured by a one-question questionnaire. Future works may use structured questionnaires (e.g., Perceived stress scale, Standard stress scale) or clinical indicators, such as pathological information (e.g., cortisol, epinephrine, norepinephrine), to measure stress [38]. Secondly, gender difference was only interpreted comprehensively and extensively in terms of social, economic, and stress coping because KNHANES dataset did not include biological indicators of gender differences, such as sex hormone concentrations. Third, the causal relationship between variables could not be determined because of the study’s cross-sectional design. Future research using objective measurements and longitudinal data is needed to prove the causal relationship between muscle strength, perceived stress, and depressive symptoms.
Discussion
- Depressive symptoms in community-dwelling older adults are a severe public health concern. Moreover, the factors related to depressive symptoms show gender differences. Therefore, this study examined the gender differences concerning muscle strength and depressive symptoms mediated by perceived stress. The findings from this study suggested that perceived stress had a mediating effect on the relation between muscle strength and depressive symptoms, but the effect differed by gender. These findings have important implications for public health nurses to develop depressive symptoms intervention for older adults. A stress-mediated depressive symptoms intervention program should be developed with consideration for females’ needs.
Conclusions
-
Conflict of interest
Ju Young Yoon is an editorial board member of the Journal of Korean Community Health Nursing. She will not involve in the review process of this manuscript. No conflict of interest has been declared by authors.
-
Funding
This work was supported by the Research Institute of Nursing Science, Seoul National University Student Research Grant in 2021, and was supported by the BK21 four project (Center for Human-Caring Nurse Leaders for the Future) funded by the Ministry of Education (MOE, Korea) and National Research Foundation of Korea (NRF). GAK received a scholarship from the BK21 education program (Center for Human-Caring Nurse Leaders for the Future).
-
Authors’ contributions
Gyeong A Kang contributed to conceptualization, data curation, formal analysis, methodology, project administration, and writing – original draft, review & editing. Jihye Shin contributed to conceptualization, methodology, and writing – review & editing. Ju Young Yoon contributed to conceptualization, writing – review & editing, supervision, and validation.
-
Data availability
Or note how data can be made available such as follows: The Korea National Health and Nutrition Examination Survey (KNHANES) were obtained from the Korea Disease Control and Prevention Agency (https://knhanes.kdca.go.kr/). More details on the data collection process are available (https://doi.org/10.1093/ije/dyt228).
NOTES
Acknowledgments
Notes. Unweighted numbers, weighted percentages (%), and chi-squared values are presented for categorical variables. Mean ± SE and t-values are presented for continuous variables. Those “not living with a spouse” include widowed, divorced, or never married individuals. “Current smoking: No” includes those who have never smoked or those who used to smoke in the past but no longer do so.
NA = not applicable.
| Variable | Muscle strength | Perceived stress | Depressive symptoms |
|---|---|---|---|
| Muscle strength | - | ||
| Perceived stress | -.072*** | - | |
| Depressive symptoms | -.186*** | .342*** | - |
Notes. All values are unstandardized. Model 7 of the PROCESS macro is used. In gender variable, male is reference. Covariance: age, residential area, education attainment, household income, marital status, current smoking, high-risk drinking, history of depression treatment, number of chronic diseases.
- 1. Hu T, Zhao X, Wu M, Li Z, Luo L, Yang C, Yang F. Prevalence of depression in older adults: A systematic review and meta-analysis. Psychiatry Research. 2022;311:114511. http://doi.org/10.1016/j.psychres.2022.114511ArticlePubMed
- 2. Soysal P, Veronese N, Thompson T, Kahl KG, Fernandes BS, Prina AM, et al. Relationship between depression and frailty in older adults: A systematic review and meta-analysis. Ageing Research Reviews. 2017;36:78–87. http://doi.org/10.1016/j.arr.2017.03.005ArticlePubMed
- 3. Cao L, Zhou Y, Liu H, Shi M, Wei Y, Xia Y. Bidirectional longitudinal study of frailty and depressive symptoms among older Chinese adults. Frontiers in Aging Neuroscience. 2022;14:791971. http://doi.org/10.3389/fnagi.2022.791971ArticlePubMedPMC
- 4. Desrichard O, Vallet F, Agrigoroaei S, Fagot D, Spini D. Frailty in aging and its influence on perceived stress exposure and stress-related symptoms: evidence from the Swiss Vivre/Leben/Vivere study. European Journal of Ageing. 2018;15(4):331–338. http://doi.org/10.1007/s10433-017-0451-2ArticlePubMedPMC
- 5. Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G. Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. The Journals of Gerontology:Series A. 2004;59(3):M255–M263. https://doi.org/10.1093/gerona/59.3.M255ArticlePubMed
- 6. Folkman S. Stress: appraisal and coping. In: Gellman MD, Turner JR, editors. Encyclopedia of Behavioral Medicine. NY: Springer New York; 2013. p. 1913–1915.
- 7. Tafet GE, Nemeroff CB. The links between stress and depression: Psychoneuroendocrinological, genetic, and environmental interactions. The Journal of Neuropsychiatry and Clinical Neuroscience. 2016;28(2):77–88. http://doi.org/10.1176/appi.neuropsych.15030053ArticlePubMed
- 8. Lazarus R, Folkman S. Stress, appraisal, and coping. NY: Springer Publishing Company; 1984. 444 p.
- 9. Cabanas-Sánchez V, Esteban-Cornejo I, Parra-Soto S, Petermann-Rocha F, Gray SR, Rodríguez-Artalejo F, et al. Muscle strength and incidence of depression and anxiety: findings from the UK biobank prospective cohort study. Journal of Cachexia, Sarcopenia and Muscle. 2022;13(4):1983–1994. http://doi.org/10.1002/jcsm.12963ArticlePubMedPMC
- 10. Severinsen MCK, Pedersen BK. Muscle-Organ Crosstalk: The emerging roles of myokines. Endocrine Reviews. 2020;41(4):594–609. http://doi.org/10.1210/endrev/bnaa016ArticlePubMedPMC
- 11. Kim SE, Kim HN, Cho J, Kwon MJ, Chang Y, Ryu S, et al. Direct and indirect effects of five factor personality and gender on depressive symptoms mediated by perceived stress. PloS One. 2016;11(4):e0154140. http://doi.org/10.1371/journal.pone.0154140ArticlePubMedPMC
- 12. Smith L, Firth J, Grabovac I, Koyanagi A, Veronese N, Stubbs B, et al. The association of grip strength with depressive symptoms and cortisol in hair: A cross-sectional study of older adults. Scandinavian Journal of Medicine & Science in Sports. 2019;29(10):1604–1609. http://doi.org/10.1111/sms.13497ArticlePubMed
- 13. Noh HM, Park YS. Handgrip strength, dynapenia, and mental health in older Koreans. Science Reports. 2020;10(1):4004. http://doi.org/10.1038/s41598-020-60835-4ArticlePubMedPMC
- 14. Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in older adults: Evidence for a phenotype. The Journals of Gerontology: Series A. 2001;56(3):M146–M157. http://doi.org/10.1093/gerona/56.3.m146ArticlePubMed
- 15. Soh Y, Won CW. Sex differences in association between body composition and frailty or physical performance in community-dwelling older adults. Medicine. 2021;100(4):e24400. http://doi.org/10.1097/MD.0000000000024400ArticlePubMedPMC
- 16. Verma R, Balhara YP, Gupta CS. Gender differences in stress response: Role of developmental and biological determinants. Industrial Psychiatry Journal. 2011;20(1):4–10. http://doi.org/10.4103/0972-6748.98407ArticlePubMedPMC
- 17. Swinkels J, Tilburg Tv, Verbakel E, Broese van Groenou M. Explaining the gender gap in the caregiving burden of partner caregivers. The Journals of Gerontology: Series B. 2017;74(2):309–317. https://doi.org/10.1093/geronb/gbx036ArticlePubMedPMC
- 18. Angrisani M, Lee J, Meijer E. The gender gap in education and late-life cognition: Evidence from multiple countries and birth cohorts. Journal of the Economics of Ageing. 2020;16:100232. https://doi.org/10.1016/j.jeoa.2019.100232ArticlePubMedPMC
- 19. Kweon S, Kim Y, Jang MJ, Kim Y, Kim K, Choi S, et al. Data resource profile: the Korea National Health and Nutrition Examination Survey (KNHANES). International Journal of Epidemiology. 2014;43(1):69–77. https://doi.org/10.1093/ije/dyt228ArticlePubMedPMC
- 20. Bobos P, Nazari G, Lu Z, MacDermid JC. Measurement properties of the hand grip strength assessment: A systematic review with meta-analysis. Archives of Physical Medicine and Rehabilitation. 2020;101(3):553–565. https://doi.org/10.1016/j.apmr.2019.10.18321ArticlePubMed
- 21. Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T, et al. Sarcopenia: revised European consensus on definition and diagnosis. Age and Ageing. 2019;48(4):601. https://doi.org/10.1093/ageing/afz046ArticlePubMedPMC
- 22. Kroenke K, Spitzer RL, Williams JB. The PHQ-9: validity of a brief depression severity measure. Journal of General Internal Medicine. 2001;16(9):606–613. https://doi.org/10.1046/j.1525-1497.2001.016009606.xArticlePubMedPMC
- 23. Kim H, Jeong W, Kim SH, Park YS, Jang SI, Park EC. Association between changes in handgrip strength and depression in Korean adults: a longitudinal panel study. Science Reports. 2022;12(1):13643. https://doi.org/10.1038/s41598-022-18089-9ArticlePubMedPMC
- 24. Hayes AF, Rockwood NJ. Conditional process analysis: Concepts, computation, and advances in the modeling of the contingencies of mechanisms. 2020;64(1):19–54. https://doi.org/10.1177/0002764219859633
- 25. Hayes AF. An index and test of linear moderated mediation. Multivariate Behavioral Research. 2015;50(1):1–22. https://doi.org/10.1080/00273171.2014.962683ArticlePubMed
- 26. Mucher P, Batmyagmar D, Perkmann T, Repl M, Radakovics A, Ponocny-Seliger E, et al. Basal myokine levels are associated with quality of life and depressed mood in older adults. Psychophysiology. 2021;58(5):e13799. https://doi.org/10.1111/psyp.13799ArticlePubMedPMC
- 27. Ashdown-Franks G, Koyanagi A, Vancampfort D, Smith L, Firth J, Schuch F, et al. Sedentary behavior and perceived stress among adults aged ≥50 years in six low- and middle-income countries. Maturitas. 2018;116:100–107. https://doi.org/10.1016/j.maturitas.2018.08.005ArticlePubMed
- 28. Davies K, Maharani A, Chandola T, Todd C, Pendleton N. The longitudinal relationship between loneliness, social isolation, and frailty in older adults in England: a prospective analysis. The Lancet Healthy longevity. 2021;2(2):e70–e77. https://doi.org/10.1016/S2666-7568(20)30038-6ArticlePubMed
- 29. Salk RH, Hyde JS, Abramson LY. Gender differences in depression in representative national samples: Meta-analyses of diagnoses and symptoms. Psychological Bulletin. 2017;143(8):783–822. https://doi.org/10.1037/bul0000102ArticlePubMedPMC
- 30. Zapater-Fajarí M, Crespo-Sanmiguel I, Pulopulos MM, Hidalgo V, Salvador A. Resilience and psychobiological response to stress in older reople: The mediating role of coping strategies. Frontiers in Aging Neuroscience. 2021;13:632141. https://doi.org/10.3389/fnagi.2021.632141ArticlePubMedPMC
- 31. Slavich GM, Sacher J. Stress, sex hormones, inflammation, and major depressive disorder: Extending social signal transduction theory of depression to account for sex differences in mood disorders. Psychopharmacology. 2019;236(10):3063–3079. https://doi.org/10.1007/s00213-019-05326-9ArticlePubMedPMC
- 32. Kendler KS, Thornton LM, Prescott CA. Gender differences in the rates of exposure to stressful life events and sensitivity to their depressogenic effects. The American Journal of Psychiatry. 2001;158(4):587–593. https://doi.org/10.1176/appi.ajp.158.4.587ArticlePubMed
- 33. Tugade MM, Fredrickson BL. Resilient individuals use positive emotions to bounce back from negative emotional experiences. Journal of Personality and Social Psychology. 2004;86(2):320–333. https://doi.org/10.1037/0022-3514.86.2.320ArticlePubMedPMC
- 34. Gopinath B, Kifley A, Flood VM, Mitchell P. Physical activity as a determinant of successful aging over ten years. Scientific Reports. 2018;8(1):10522. https://doi.org/10.1038/s41598-018-28526-3ArticlePubMedPMC
- 35. Goldfarb EV, Seo D, Sinha R. Sex differences in neural stress responses and correlation with subjective stress and stress regulation. Neurobiology of Stress. 2019;11:100177. https://doi.org/10.1016/j.ynstr.2019.100177ArticlePubMedPMC
- 36. Lundberg U. Stress hormones in health and illness: the roles of work and gender. Psychoneuroendocrinology. 2005;30(10):1017–1021. https://doi.org/10.1016/j.psyneuen.2005.03.014ArticlePubMed
- 37. Holvast F, Massoudi B, Oude Voshaar RC, Verhaak PFM. Non-pharmacological treatment for depressed older patients in primary care: A systematic review and meta-analysis. PloS One. 2017;12(9):e0184666. https://doi.org/10.1371/journal.pone.0184666ArticlePubMedPMC
- 38. Cohen S, Kamarck T, Mermelstein R. A global measure of perceived stress. Journal of Health and Social Behavior. 1983;24(4):385–396. https://doi.org/10.2307/2136404ArticlePubMed
REFERENCES
Figure & Data
References
Citations


 KACHN
KACHN
 PubReader
PubReader ePub Link
ePub Link Cite
Cite